Easy integration
Deploy QB Control on our modules for turnkey performance, or implement it atop existing equipment from other vendors through retrofit kits (signal adapters, I/O expansions, panel upgrades) and software connectors (e.g., OPC UA, REST, MQTT) to protect past investments.
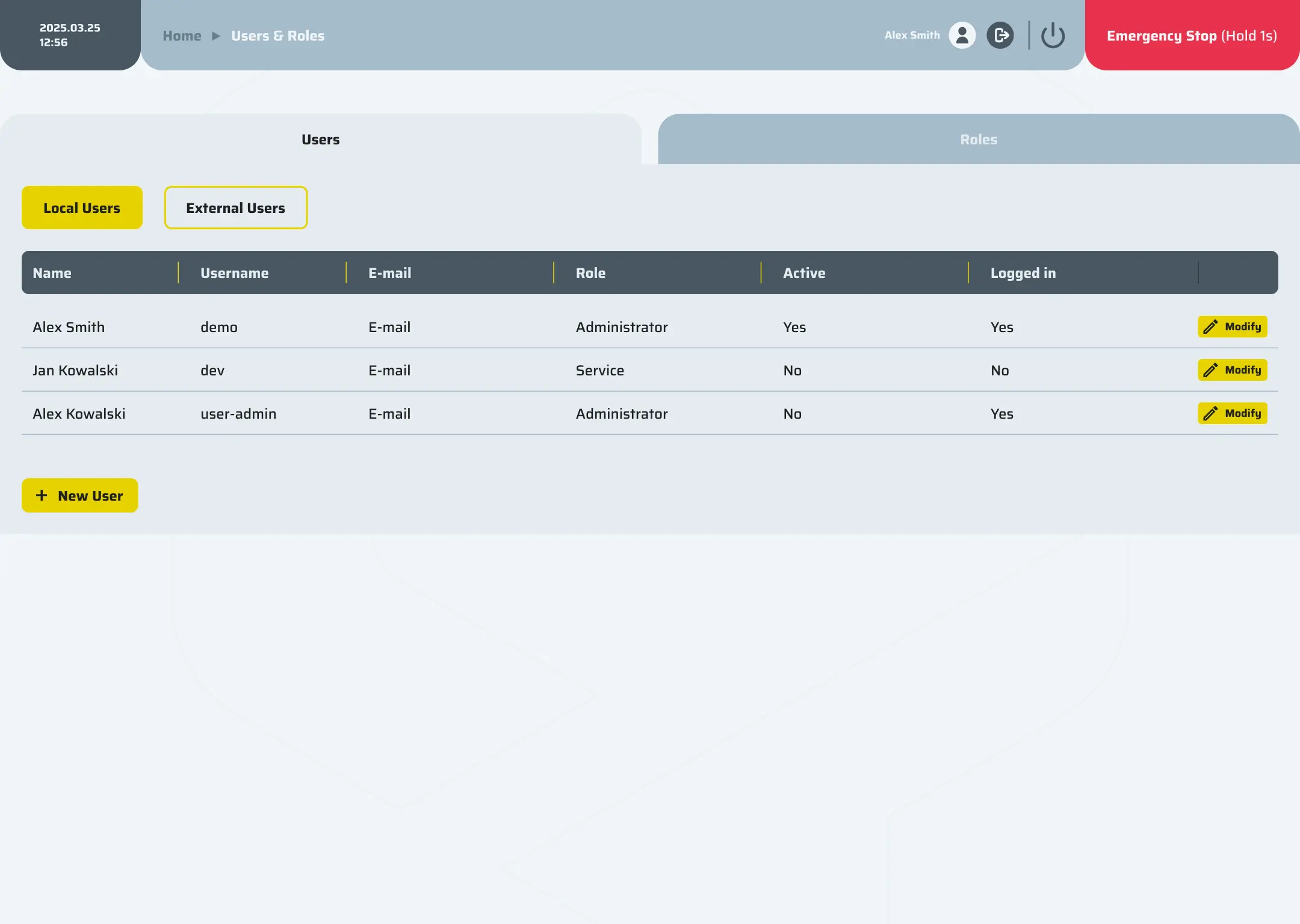
Data integrity
A streamlined system ensures compliance, security, and efficiency through an integrated audit trail, e-signatures, role-based access, versioning, calibration records, and seamless export options to PDF or CSV formats.
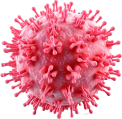
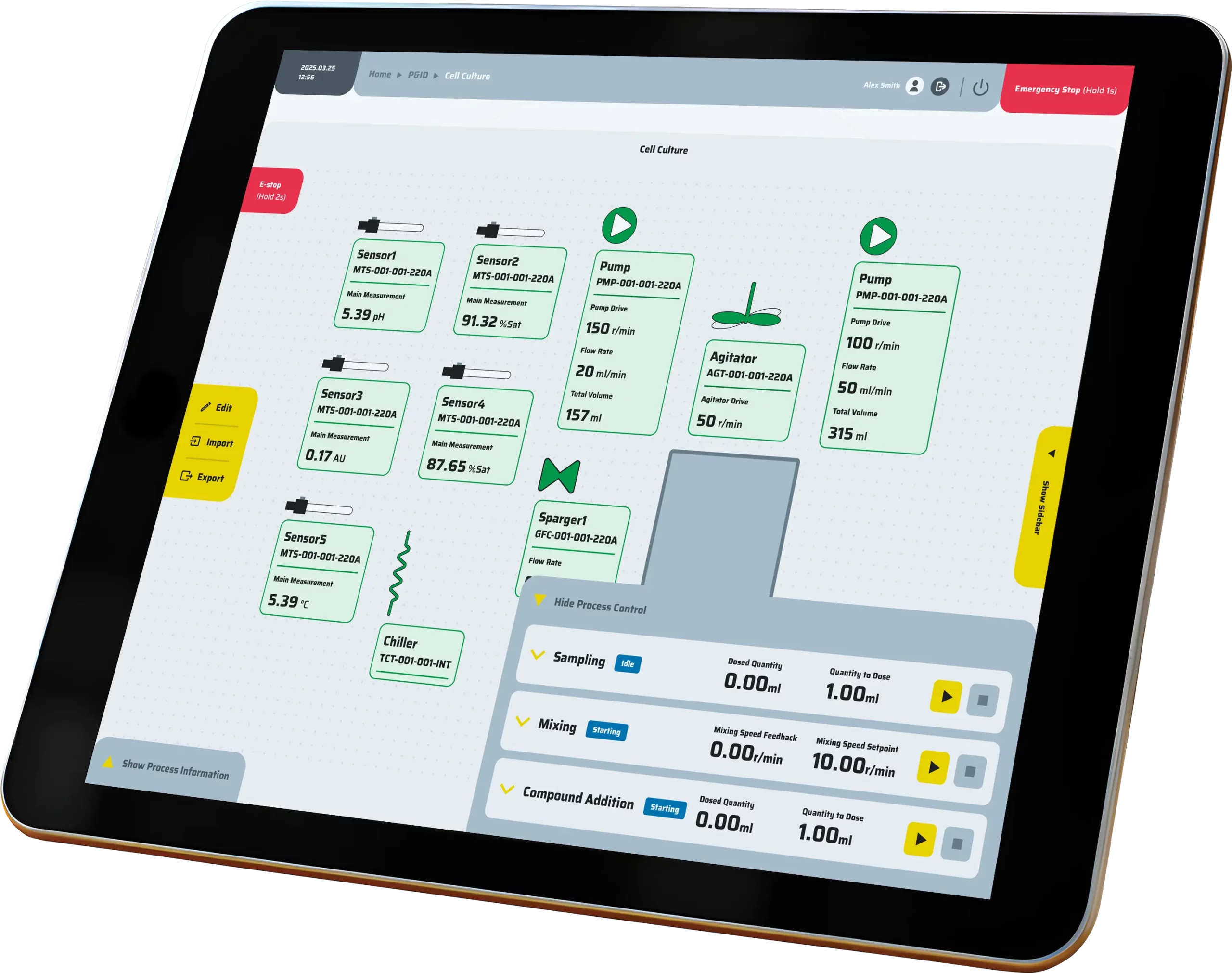
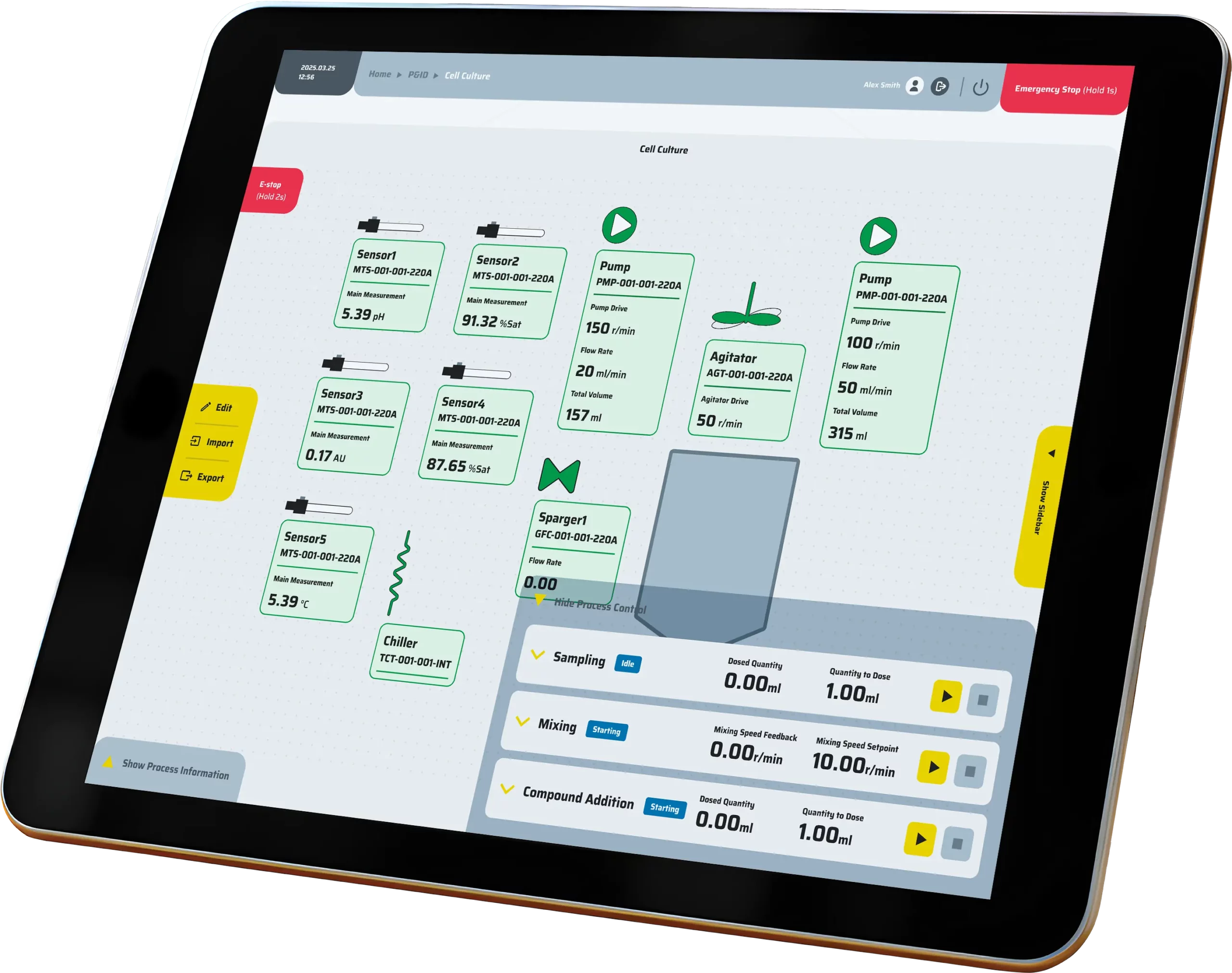
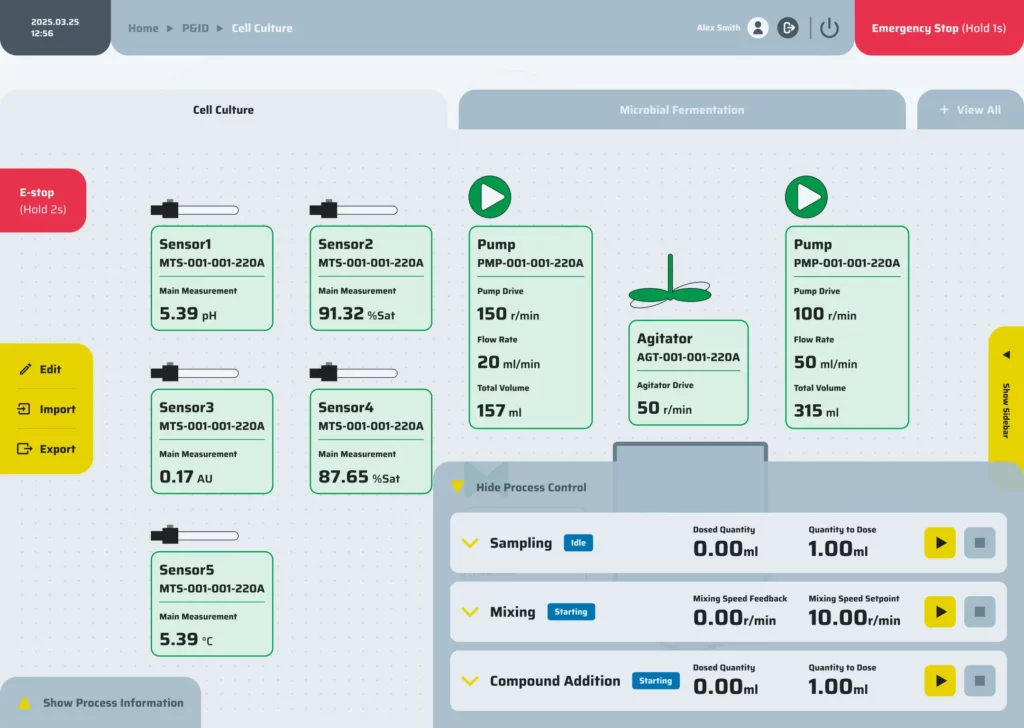
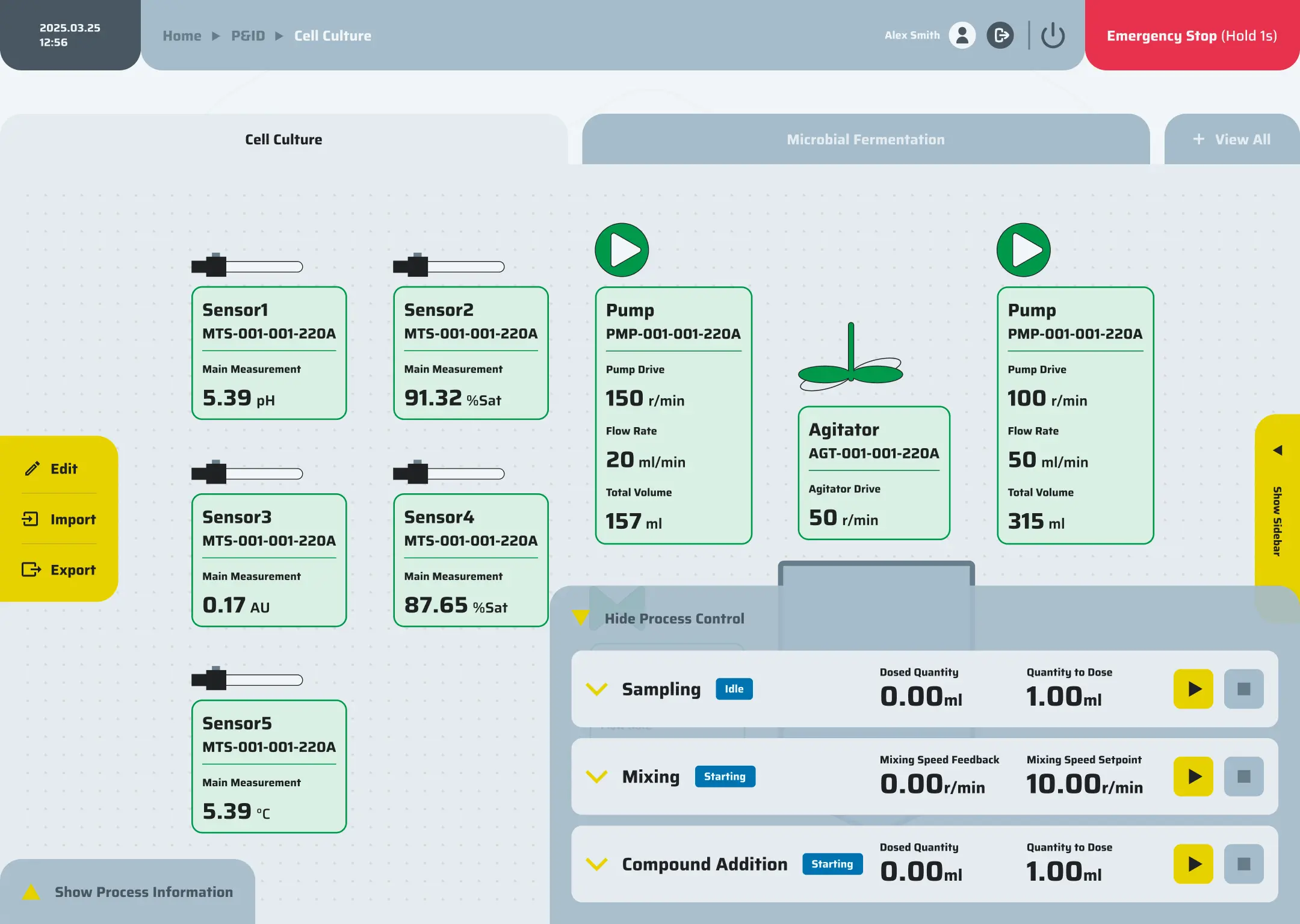
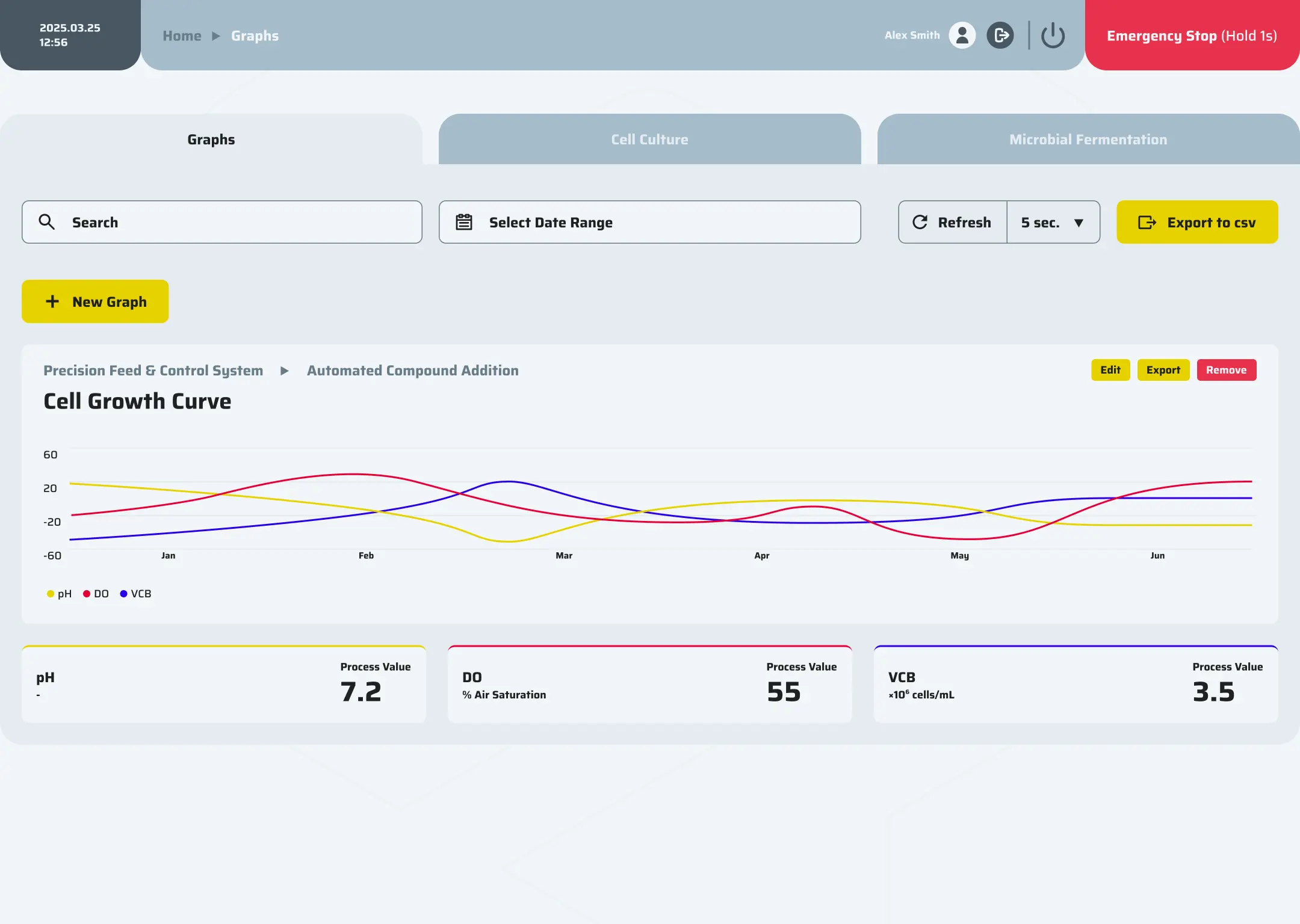
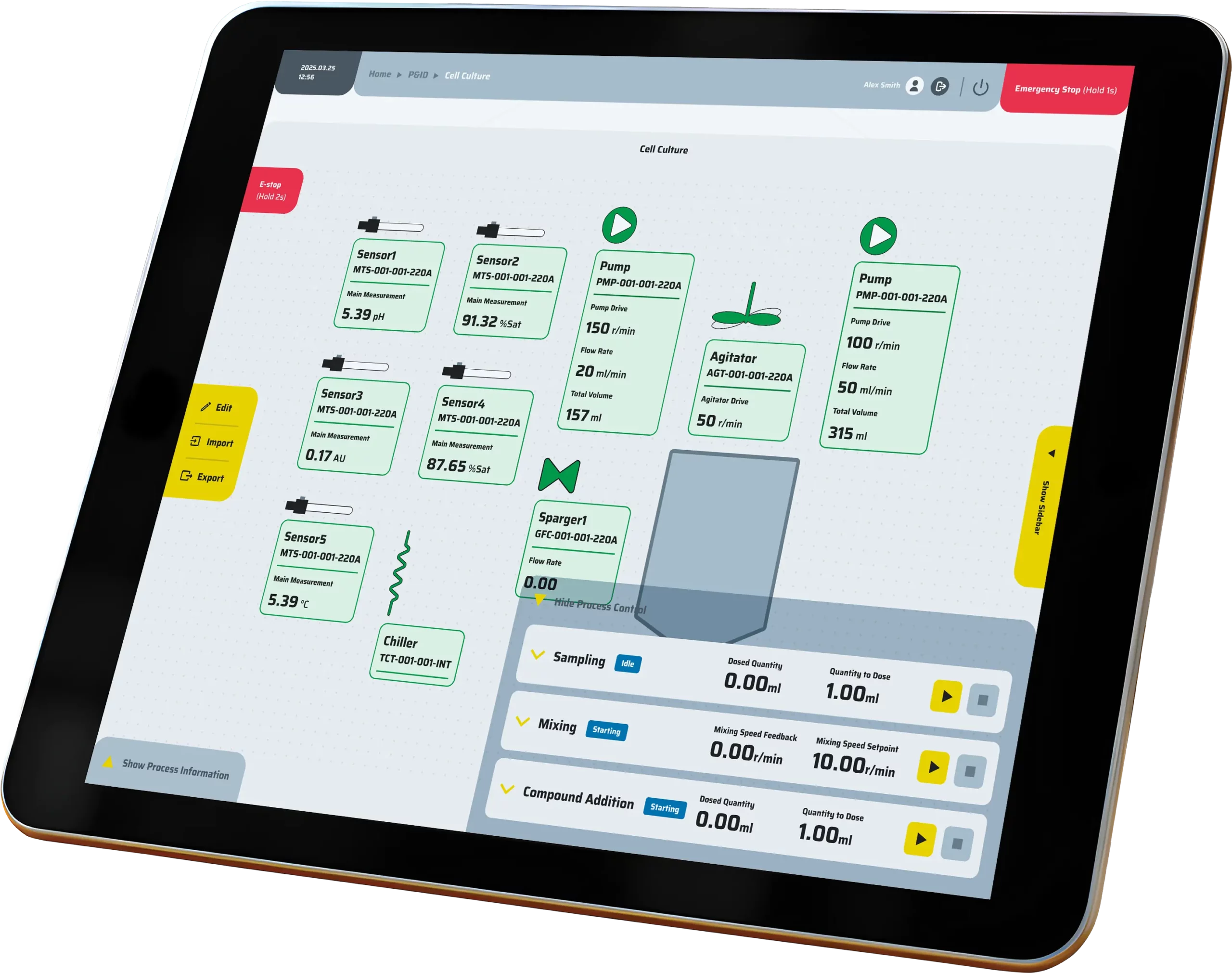
Precision control
Precision control is achieved through the integration of P&ID loops, event-based logic, cascades, and hardware interlocks, all governed by comprehensive alarm policies to ensure safe and accurate operation.